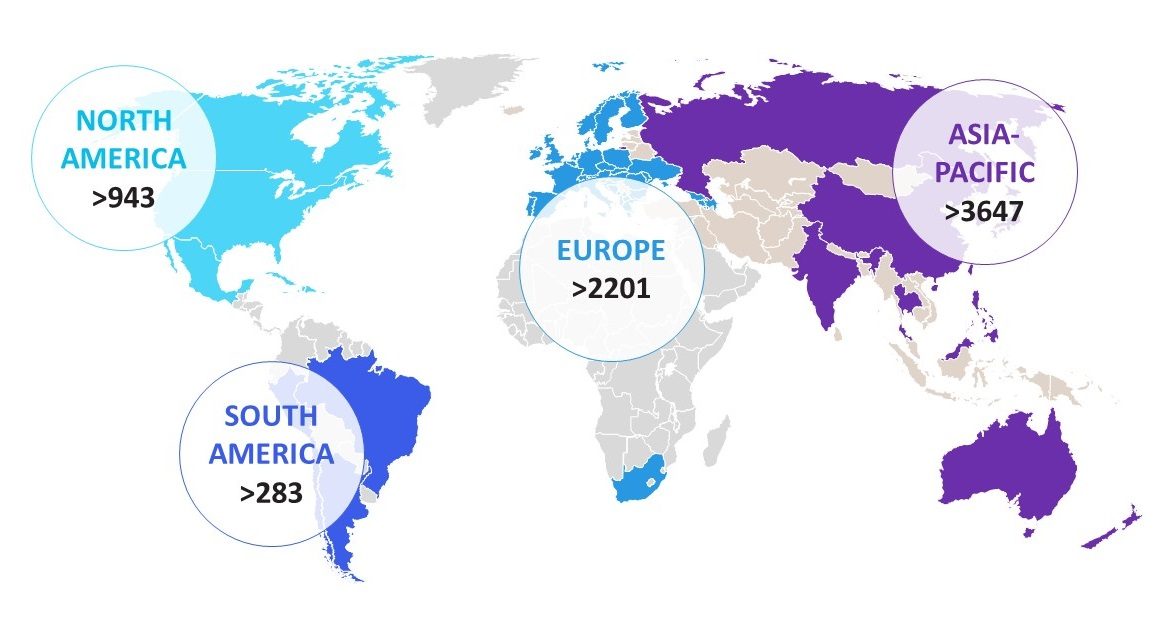
Supplement your clinical operations program with an experienced partner at your side. As a clinical service provider, our team at Labcorp has been engaged in global, scalable Functional Service Provider (FSP) relationships with biopharmaceutical clients for more than 30 years across 60+ countries. We know how to serve as a vital extension of your resources across a number of functions—including clinical monitoring, drug safety, regulatory and clinical trial management—all aligned to your culture, mission and business goals.
Our engagements are customized to your needs—from a few dedicated full-time equivalent (FTE) staff to 150+ FTEs across several functional areas. Project-managed by you, employed by Labcorp–we provide the specialized staff you need for the following job functions:
- Clinical Research Associates (CRAs)
- Project Managers
- Program Leaders
- Data Managers
- Data Coordinators and Programmers
- Medical Writers
- Regulatory Staff
- Biostatisticians
- Clinical Trial Leads (CTLs)
- Clinical Trial Managers (CTMs)
With our resource menu at your disposal, you can quickly ramp up or down your staffing levels—without focusing on recruitment and retention—and optimize your cost distribution. We continually invest in attracting and retaining the right talent, working with people who can integrate into your teams and deliver seamless business continuity.
Our team is here to help deliver confidence to your team. By starting with consistent delivery of flexible resources and functional services, we believe in being more than a partner. In fact, we often foster long-term, productive relationships that last more than 10 years. Our focus is centered on your needs as we continue to invest in building, training and retaining our workforce on your behalf, with a focus on innovation, flexibility and cost competitiveness.
Throughout every interaction, we implement rigorous and visible Key Performance Indicators (KPIs) as core components of our quality control metrics to enable the transparency and accountability you need to move forward.
Tailor our FSP delivery model to your unique requirements
Embedded Employee:
Input-based model |
Enhanced FTE:
Input-based model |
Unit Deliverable:
Output-based model |
|---|
You retain overall accountability, responsibility and direction of the study— including provision of direct management and work assignments to your assigned resources.
|
You retain overall accountability, responsibility and direction of the study, while our team at Labcorp is responsible for liaising with you and making work assignments directly with your assigned resources.
|
We manage the day-to-day tasks and the resources assigned to your program. We retain responsibility for decisions concerned with hiring and line management of our employees who will work from Labcorp offices or who are field-based. The staff members assigned can use our tools and systems or yours.
|