Author: Erin Trachet, Sr. Scientific Advisor, Oncology / Sr. Manager, Proposal Development
Date: December 2016
Acute myeloid leukemia (AML) is a malignant disorder of the progenitor cells in myeloid hematopoiesis and represents a genetically heterogeneous cancer. The onset of AML is thought to require cooperation between active proliferation and defects in myeloid differentiation, which often results in chromosomal translocation (Gilliland et al., 2004). The annual incidence of AML is ~1.8 per 100,000 people with incidence increasing significantly with age. Clinically, a number of novel therapeutic strategies to improve outcomes in AML are being investigated. However, even with the advances in therapies and improvements in early diagnosis, the majority of patients will die from their disease.
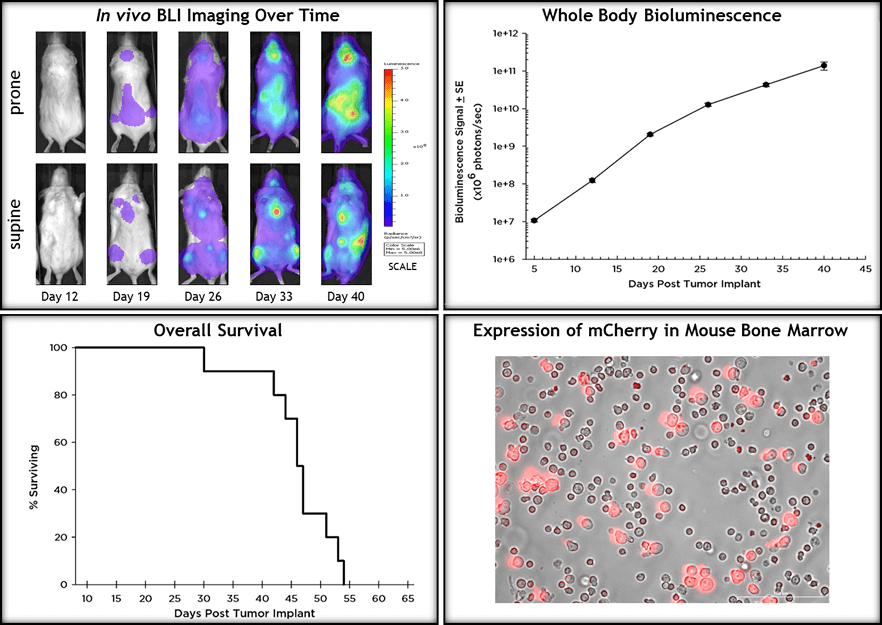
In an effort to more effectively evaluate treatments preclinically, Labcorp offers a human AML model, MV(4;11) (FUW-Luc-mCherry-puro), that has been modified to include both luciferase and mCherry reporter constructs. Through expression of luciferase, bioluminescent imaging (BLI) can be used to quantitatively measure and track in vivo disease progression in a longitudinal manner. In addition, expression of mCherry can be used ex vivo to detect presence or absence of tumor cells in the bone marrow. At Labcorp we have optimized the MV(4;11) model in female NSG mice (Jackson Labs). Our ability to track disease progression with BLI makes this model a powerful tool for investigators discovering and developing novel drugs for AML by coupling traditional pharmacology endpoints such as overall survival and body weight loss with in vivo, quantitative disease progression over time. Preclinically, BLI has become the cornerstone imaging modality in the quantification of hematologic malignancy models and is invaluable to the drug discovery process.
Contact Labcorp today for more Information on the MV(4;11) (FUW-Luc-mCherry-puro) model or one of our other hematologic malignancy models—we have dozens of cell lines!
References
Connect
Let's start a conversation
Contact Us