Author: David Draper
Date: March 2020
Enhancement of the tumor antigen-specific response is an intended outcome of immunotherapy. Be it through an acute increase in anti-tumor immunity or formation of long-lived immunological memory, drugs that successfully boost the host adaptive immune response have a better chance of clinical success1.
While numerous assays are available to measure antigen specificity, many of these have limitations as they necessitate the use of model-specific reagents, genetically engineered animals, or cell subset purification steps. Our Preclinical Oncology offers a simplistic and affordable solution through ELISpot/FluoroSpot analysis.
ELISpot and FluoroSpot use sandwich ELISA-based technology to measure the frequency of cells that produce and release soluble targets. Commercial kits are available to measure targets that include cytokines, chemokines, immunoglobulins, growth factors, and more. We offer single color enzyme-linked immuno-spot analysis of one target (ELISpot) as well as multi-color fluorescence-based analysis of up to 3 targets simultaneously (FluoroSpot). A schematic that illustrates the FluoroSpot procedure for the analysis of tumor-specific IFNγ and IL-4 producing cells is shown in Figure 1.
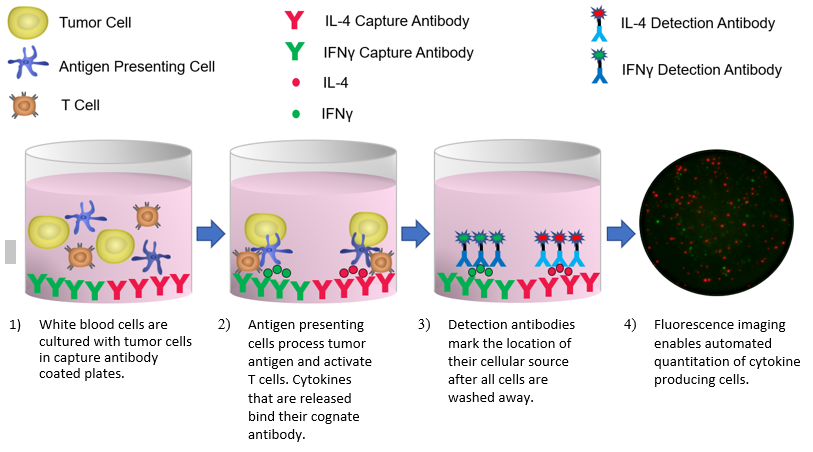
Figure 1: ELISpot/FluoroSpot Procedure
In this Technology Spotlight, we demonstrate application of ELISpot/FluoroSpot in preclinical immuno-oncology research using the Hepa1-6-luc murine hepatocellular carcinoma model (Figure 2). To this end, using FluoroSpot, we measured tumor-specific immunity in the peripheral blood of tumor bearing mice and show that in vivo checkpoint inhibition with anti-PD-1 treatment increases the number of circulating tumor-specific cells producing IFNγ. Furthermore, anti-PD-1 shifts the IFNγ/IL-4 cytokine profile from an immunosuppressive response towards a more pro-inflammatory and anti-tumor signature.
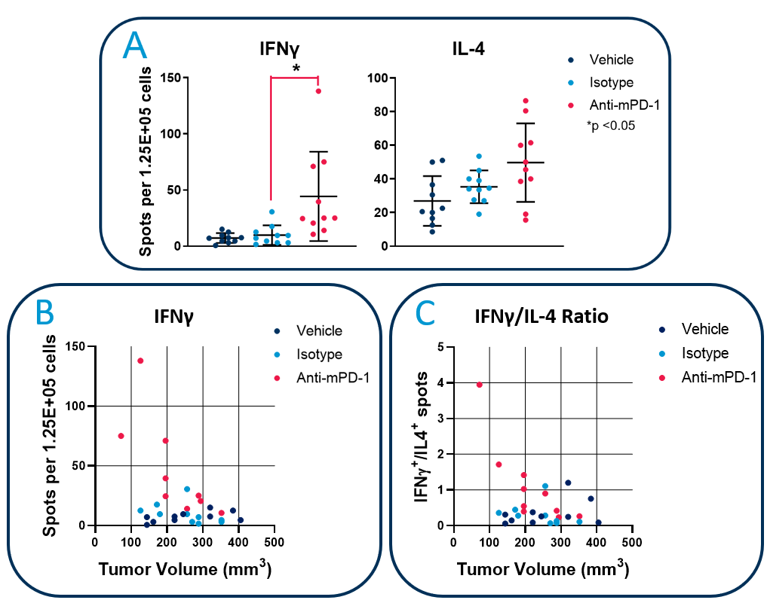
Figure 2: FluoroSpot analysis of anti-tumor immunity in Hepa1-6-luc tumor bearing mice. C57BL/6 mice with established Hepa1-6-luc tumors were dosed with anti-PD-1 or isotype control antibodies. White blood cells from 200µL of peripheral blood were cultured with irradiated Hepa1-6-luc cells. A) Quantitation of the frequency of IFNγ and IL-4 secreting cells using a CTL ImmunoSpot® S6 Universal analyzer. B) Analysis of the IFNγ-producing cell frequency vs tumor volume in each mouse. C). Th1/Th2 balance in each mouse assessed by measuring the ratio of IFNγ/IL-4 producing cells.
Similar to the mechanism of action for a vaccine, a growing tumor elicits an adaptive immune response that consists of antibody and cytokine producing tumor-specific cells1. The quality of the response can be characterized by examination of IFNγ and IL-4. IFNγ has been well documented to inhibit tumor growth by enhancing T cell-mediated by cytotoxicity. In contrast, IL-4 promotes tumor growth through a variety of immunosuppressive effects. This dichotomy is often referred to as a Th1/Th2 balance2. As shown in Figure 2, anti-PD-1 increased the frequency of tumor-responsive cells producing IFNγ following ex vivo stimulation. In contrast, the frequency of IL-4 producing cells was not significantly increased, suggesting checkpoint inhibition triggered an enhancement of anti-tumor protective immunity. In support of this, mice with the highest frequencies of IFNγ producing cells harbored tumors with the smallest volumes. Furthermore, anti-PD-1 increased the ratio of IFNγ/IL-4 producing cells, which again was most pronounced in mice with the smallest tumors.
Taken together, this dataset demonstrates how ELISpot/FluoroSpot can further reveal drug effects on the tumor-specific immune response. Moreover, because the assay can be performed with small volumes of blood, ELISpot/FluoroSpot can be applied to efficacy arms longitudinally. Thus, opportunity to gain mechanistic insight on drug activity without adding a second sampling arm to the study is provided. For more information on how ELISpot/FluoroSpot can be applied to your preclinical oncology and immuno-oncology research, contact the scientists at Labcorp Preclinical Oncology.
References
Connect
Let's start a conversation
Contact Us